Magnetic resonance imaging
of the normal infant brain:
term to 2 years
Frances M Cowan
Chapter Contents
- Introduction
- Pulse sequences used by our group for imaging neonates and young infants
- MR imaging of the brain in the term infant: conventional T1 and T2 weighted sequences
- Contrast enhancement
- Diffusion weighted imaging of the neonatal brain
- MR changes in the cerebrum, brain stem and cerebellum occuring after term:
conventional T1 and T2 weighted sequences - Observations of other areas of the brain seen on MR brain scans from term to years
- Functional MR imaging
- Summary
- Abbreviations
- References
Introduction
Magnetic resonance (MR) imaging of the infant brain has given an enormous insight into the maturational processes that take place after birth. The technique has made it possible to see in minute detail changes in cortical folding, involution of the germinal layer, premyelination changes within white matter, myelination, iron deposition, and the growth of different regions of the brain that is not possible with computed tomography or ultrasound.
Establishing normality is especially difficult because the appearance of the normal brain is changing almost weekly. A large number of infants need to be studied with different strength magnets and using different sequences to fully appreciate the range of normal appearances4–8, 11, 19, 25, 28, 37–39, 41, 49, 53, 54, 58, 59, 61, 62, 64, 71, 78–81, 86, 88, 91 . The introduction of new sequences often means that new norms need to be established. Centers must determine normal appearances for different ages on their own system.
< prev | top | contents | next >
Pulse sequences used by our own group for imaging neonates and young infants
We have a 1 Tesla Picker HPQ Vista/Plus system and a 1 Tesla novel short small bore system which is situated in the neonatal intensive care unit. For imaging and multinuclear spectroscopy we have a 1.5 Tesla Picker Eclipse system. All three scanners are used for imaging infants up to term. After term, the neonatal unit magnet cannot be used as the bore is too small57. Our MR scans are not angled as steeply as is usual for computed tomography (CT). This makes the appearance of images, particularly the basal ganglia, thalami and internal capsule, different from some published data. These differences must be taken into account when interpreting images. Details of the sequences used on the different systems at different ages are given in Table 4.1. The slice thickness we use is 4–6mm with no slice gap.
An excellent review of different pulse sequences and their relative merits in the pediatric context is given by Barkovich8.
Table 4.1 Pulse sequences. Term to 3 months
| 1T Picker HPQ | 1T NNU magnet | 1.5 T Eclipse | |||||
|---|---|---|---|---|---|---|---|
| TR/TE | FOV | TR/TE | FOV | TR/TE | FOV | ||
| T1W SE | 860/20 | 24 | 600/20 | 20 | 500/15 | 30 | |
| T2W SE | 2700/120 | 22 | |||||
| T2W FSE | 3500/208 | 20 | |||||
| Dual Echo | 2500/20–80 | 25 | |||||
| Dual FSE | 4200/15–210 | 30 | |||||
| Volume | 23/6 | 26.5 (1.6 slice thickness) | |||||
| DWI | Cardiac gated, pulsed gradient | ||||||
| SE pulse interval/200ms | |||||||
| b = 600s/mm2 | |||||||
| 4 data acquisitions | |||||||
| 2/3 perpendicular planes | |||||||
| Multislice | Single shot echoplanar | ||||||
| b = 1000 s/mm2 | |||||||
| 1 data acquisition | |||||||
| 3 perpendicular planes | |||||||
| TR6200/100 | |||||||
| matrix 100 x 100 | |||||||
| TR/TE/TI | FOV | TR/TE/T | FOV | ||||
| IR | 3800/30/950 | 22–24 | 3697/36/950 | 20 | |||
| FLAIR | 8142/150/2100 | 25 | |||||
| Sequences used after 3 months | |||||||
| IR 3 months – 2 years | 3400/30/800 | 22–24 | |||||
| >2 years | 3100/30/700 | 22–24 | |||||
| NNU, neonatal unit. | |||||||
< prev | top | contents | next >
MR imaging of the brain in the term infant: conventional T1 and T2 weighted sequences
THE CORTEX
The brain of the term (37–42 weeks) infant is similar to that of the adult in terms of its cortical folding. By 38 weeks all the sulci are formed though they become deeper over the following few weeks1 (Figs 4.1 and 4.5).
The signal intensity (SI) of the cortex is high on T1 weighted (T1W) (Figs 4.1 and 4.4a) and low on T2 weighted (T2W) images (Figs 4.2, 4.3, 4.4c and 4.5). In general, the more heavily the images are T1 weighted the higher the relative SI of the cortex (Fig. 4.1) and the more heavily they are T2 weighted, as seen with the fast spin echo (FSE) sequence (Fig. 4.3), the lower the relative SI. Highest signal contrast is seen around the Rolandic or central sulcus, that is the posterior cortex of the precentral gyrus and the anterior cortex of the postcentral gyrus (Figs 4.1–4.3). This feature remains with decreasing conspicuity for up to 2 months on T1W images (Fig. 4.8) and for up to 6 months on T2W images (Fig. 4.12).
As this pattern of SIs is the same as for myelinated white matter (WM) it has been suggested that the cortical signal in the perirolandic region is due to myelination. However, this seems unlikely for a number of reasons53. In myelinating WM T1 shortening precedes T2 shortening, whilst in the perirolandic cortex T2 shortening is more prominent and lasts longer. Also there is very little myelin in this tissue at this age. It is seen microscopically from 35 weeks’ gestation in the pre- and postcentral gyri but macroscopically only from 40 weeks in the subcortical WM, and not in the cortex. Microscopically, there is slightly more myelin in the precentral than postcentral cortex whilst the MR SIs of the two regions are the same. Finally, myelination increases enormously over the first 6 months after birth at term, during which time the signal differences in this part of the cortex become less obvious.


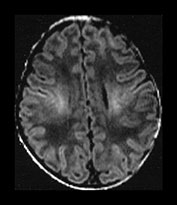
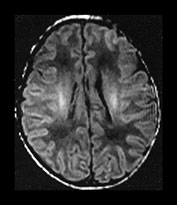

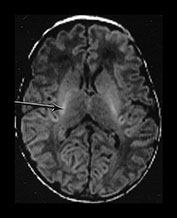
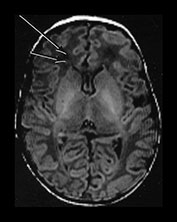



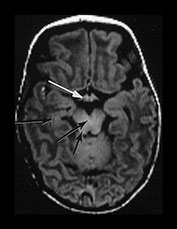
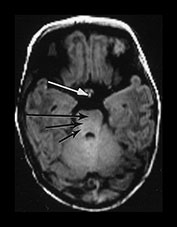
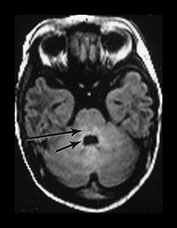

Fig. 4.1 Serial axial IR (IR 3800/30/950) images of an infant at term from high CS level to mid-cerebellar – low temporal lobe level. (b) Short T1 in the perirolandic cortex (short arrow) and central myelinated WM (long arrow). (f) Myelinated PLIC. (g) Short T1 anterior ‘caps’ (short arrow) and long T1 anterior ‘region’ (long arrow). (h) Short T1 in the GP (short b/w arrow) and the VLNT (long b/w arrow), also seen in (g). The high SI in the GP can be marked up to 42 weeks gestational age. The region of the optic radiation (white arrow) consist of three bands of low and high SI. (i) Characteristic hair pin appearance of the junction of the basal ganglia and mesencephalon with the GP anteriorly (long b/w arrow), unmyelinated tracts in the internal capsule (medium b/w arrow) and subthalamic nucleus posteriorly (short b/w arrow). The superior colliculi are seen (short white arrow) and the site of the medial geniculate body is seen laterally (long white arrow). (j) At mesencephalic level the largely unmyelinated crus is seen (long b/w arrow), as are the red nuclei (medium b/w arrow) and the medial longitudinal fasciculi (short b/w arrow). Vascular spaces are seen (short white arrow), as is the vermis of the cerebellum (long white arrow). (k) The inferior colliculi are clearly seen (short b/w arrow), as is the optic tract (white arrow). The central high SI within the brain stem is the decussation of the superior cerebellar peduncles (medium b/w arrow). The hippocampus is of intermediate SI (long b/w arrow). (l) The ventral upper pons is largely unmyelinated but of mixed SI from the pyramidal tracts (long b/w arrow). The dorsal pons is of high SI. The medial lemniscus (medium b/w arrow) and superior cerebellar peduncle (short b/w arrow) are marked. The pituitary stalk is well seen (white arrow). (m) At mid-pons level the medial lemniscus (long arrow) is well seen and the superior cerebellar peduncle (short arrow) radiates posteriorly. (n) In the lower pons the medial lemniscus (long b/w arrow), inferior cerebellar peduncle (medium b/w arrow) and dentate nucleus (short b/w arrow) are marked. The pituitary gland is seen (white arrow).
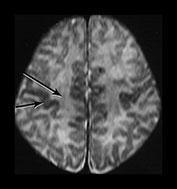


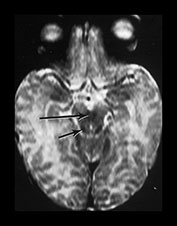
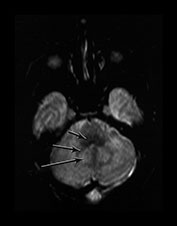
Fig. 4.2 Axial T2W (SE 2700/120) images of an infant at term. (a) Low SI in the perirolandic cortex (short arrow). The central WM is of slightly lower SI (long arrow) than the more frontal and posterior WM consistent with the onset of myelination. (b) At mid-basal ganglia level the PLIC has a small region of low SI (medium b/w arrow), the GP is of intermediate to high SI (black arrowhead) but the posterior putamen (long b/w arrow) and the VLNT (short b/w arrow) are of low SI. There is an ill-defined ‘arrowhead’-shaped region of high SI in the posterior periventricular WM (black arrow). (c) At low basal ganglia level a portion of the PLIC has a characteristic low SI globular appearance (short b/w arrow) high SI around it; small regions of high SI are seen within the ALIC (long black arrow). The VLNT is clearly demarcated and of low SI (medium b/w arrow). The posterior thalami are of intermediate SI (long b/w arrow). The optic radiations appear to consist of three tracts (short black arrow). (d) In the upper brain stem the superior colliculi (short arrow) and the medial longitudinal fasciculi (long arrow) are of low SI. (e) The medial lemnisci (short arrow) and inferior cerebellar peduncles (medium arrow) are of low SI and the dentate nuclei (long arrow) is of high SI with a low SI rim.



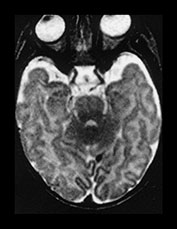
Fig. 4.3 T2W fast spin echo (SE 3500/208) images from a term infant. (a) The cortex is of lower signal than on conventional T2W images. (b) Remains of ‘caps’ are seen anterior to the anterior horns (short b/w arrow), posterior ‘arrowheads’ are more obvious (long b/w arrow) than on the conventional T2W image in Fig. 4.2 and the corpus callosum is of low SI (medium b/w arrow) despite not being myelinated at this age. Remnants of the germinal matrix are seen posterior to the thalamus (white arrow).



Fig. 4.4 Coronal views to show the myelinated PLIC (T1W SE images, a) as well as the superior and inferior colliculi and tectum (T1W, b and T2W SE images, c). The inferior colliculi myelinate before the superior colliculi. The inferior cerebral peduncle is also seen to be myelinated on both images.
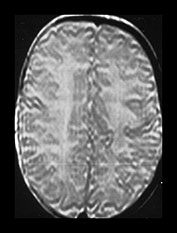
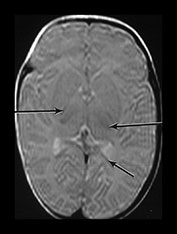

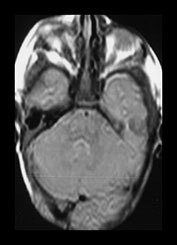
Fig. 4.5 Axial proton density (TR 2500, TE 20ms) images from an infant at term through (a) the centrum semiovale; (b) the basal ganglia; (c) the mesencephalon; (d) the cerebellum. The cortex, myelinated WM (PLIC, long arrow), VLNT (medium arrow) and the corpus callosum (short arrow) are of lower SI than the remaining central gray matter and all are of lower SI than unmyelinated WM. The CSF is of high SI. The contrast in the images is inferior to that of T1 and T2 weighted SE images.
Alternative explanations for the signal appearance of the perirolandic cortex are that it is due to more advanced development of neurons and oligodendroglia as well as greater density of synapses and dendrite formation. All of these may result in decreased free water content and increased lipid content of this particular part of the cortex9, 20-22 . If the water content decreases faster than lipid is laid down then T2 shortening would precede T1 shortening (see below for myelination). Others have suggested that these cortical signal differences are due to the paramagnetic effects of trace elements but this has not been substantiated40. Capillary proliferation or changes in protein layering15 may also account for the difference.
< prev | top | contents | next >
THE GERMINAL MATRIX
The germinal matrix, which gives rise to the cortex, largely involutes by 37 weeks but occasionally remnants of it are seen in the caudothalamic notch and posterior to the thalami at their junction with the optic radiation. Residual germinal matrix at these sites is best seen on FSE T2W sequences (Fig. 4.3). Remnants are also seen in the periventricular WM anterior to the anterior horns18, 57, 89, 54 . In older subjects this region shows patchy interruption of the ependyma, subependymal gliosis and finer myelin than in adjacent WM. This zone corresponds to the subcallosal fasciculus and it is thought that defects in the ependymal lining appear in this region during normal development as the germinal matrix disappears30, 54.
WHITE MATTER
In contrast to the relative completeness of the cortical folding, myelination which has been fairly static and confined to the brain stem, globus pallidus (GP) and ventro-lateral nucleus of the thalamus (VLNT) up to term16, 29, 34, 50, 83, 92 , shows an enormous spurt from about 38 weeks’ gestation onwards. This continues over the first year, and to a lesser extent into the second year and even to a small degree, into adolescence.
Myelin is composed of a bilayer of lipids with several large proteins traversing the bilayer. These include myelin basic protein and proteolipid protein. Histologically, myelination starts at about 25 weeks’ gestation in the GP, VLNT and some tracts in the posterior limb of the internal capsule (PLIC)16, 32, 34, 36, 50 , 10–12 weeks before it is seen in WM tracts on MR images. By 32 weeks myelin is seen microscopically in the ascending thalamocortical tracts and the descending corticospinal tracts and by 35 weeks in the striatum, and pre- and postcentral gyri. By 37 weeks it is apparent in the anterior limb of the internal capsule (ALIC) and optic radiation. At term, myelin is seen macroscopically in the thalamus, ALIC and PLIC, part of the GP, optic and acoustic radiations as well as in the pre- and postcentral subcortical WM. The pattern of myelin seen at term with MR imaging is described in Table 4.2.
< prev | top | contents | next >
Table 4.2 Regions of the brain that are myelinated on MR images at 37–42 weeks
| Centrum semiovale | Central portion |
| Visual system | Optic nerves and tracts Beginning of the optic radiations |
| Basal ganglia | Globus pallidus (partly) |
| Thalami | Ventro-lateral nucleus |
| Internal capsule | Posterior third |
| Brain stem and | Medial and lateral leminisci |
| mesencephalon | Medial longitudinal fasciculi |
| Inferior and superior cerebellar peduncles | |
| Decussation of the superior cerebellar peduncles | |
| Inferior colliculi | |
| Cerebellum | Peduncles |
| Dentate nucleus (partly) |
The degree to which myelin is seen histologically depends on the stains used36. Stains for myelin basic protein show myelin earlier than those that select more for lipid content. It is well recognized that myelin appears earlier and proceeds faster on T1W images than it does on conventional T2W SE images. This difference in timing of the appearance of myelination between the two sequences may be due to the difference in water binding resulting from the process of myelination. The shortening of T1 is temporally related to the increase in cholesterol and glycolipids that is associated with the formation of myelin from oligodendrocytes. The shortening of T2 is associated with the tightening of the spiral myelin around the axon, changes in myelin protein and saturation of polyunsaturated fatty acids in the myelin membrane. These processes are hydrophilic and alter the ratio of bound to free water in the different layers of the myelin6, 8, 88 .
The internal capsule
On T1W images the PLIC should be myelinated at term (Fig. 4.1 and Fig. 4.4) and absence of the short T1 appearance has great significance for future neurodevelopment 27, 63, 74, 75, 77 . Myelination in the PLIC is best seen on inversion recovery (IR) images where the signal is more extensive in length and breadth and the contrast with the surrounding tissues is greater than with T1W SE images.
In the term infant the PLIC starts to myelinate around 37 weeks. The increase in signal in the PLIC over the next 1–3 weeks is a constant feature that is prognostically very important (see above). It is our experience that the PLIC myelinates a little earlier in infants born very preterm where it often appears by 35 weeks. It is therefore necessary when reporting the stage of myelination of the PLIC to be certain of the infant’s gestational age and postnatal age, if born very early.
On conventional T2W SE images at low basal ganglia level only a small region in the most posterior portion of the PLIC appears myelinated (i.e. of short T2). It has a characteristic globular appearance (Fig. 4.2b,c) and is surrounded almost entirely by signal of intermediate intensity that separates it from the putamen and thalamus. On FSE images the region of short T2 is longer (Fig. 4.3b) and may be as extensive as the high SI on the T1W images.
The ALIC is often clearly seen and is of low SI on T1W (Fig. 4.1g,h) and intermediate SI on T2W images with small areas of high SI within the line of the capsule (Fig. 4.2b and Fig. 4.3b). These areas have the appearance of small cysts and may be due to vascular spaces; their aetiology is not clear.
< prev | top | contents | next >
Table 4.3 Unmyelinated white matter on MR images at term
| Most WM within the cerebral hemispheres |
| Anterior 2/3 of the PLIC |
| ALIC |
| External capsule |
| Corpus callosum |
| Anterior brain stem |
Unmyelinated white matter in the hemispheres
The distribution of unmyelinated WM is given in Table 4.3. Within the unmyelinated WM which is generally of intermediate to low SI on T1W and of high SI on T2W images there is some variation (Figs 4.1–4.3). Around the anterior horns are small ‘caps’ of low SI on T2W images that are probably migrating cells or remnants of them originating in the germinal matrix (see section on the germinal matrix) 18, 30, 55, 57, 89 . Adjacent, but more peripherally is an arrow-shaped region of high SI on T2W images and low SI on T1W images. This is relatively cell-poor on histology33. These regions are better seen on T1W and T2W FSE images than conventional T2W SE images. Posterior to the occipital horns on axial views (Figs 4.1–4.3) and above the ventricles on coronal views, there are similar regions shaped like ‘arrowheads’ that stand out from the surrounding WM. These features are much more prominent earlier in gestation and should not be obvious at term 18, 57, 89 . Indeed ‘caps’ are only obvious at term if the surrounding WM has an abnormally long T1 and T2. The ‘arrowheads’ should not be well defined at term even if the periventricular WM is of slightly long T1 and T257.
The WM should not be of uniform SI but change slightly at it goes towards the cortex so that the SI becomes closer to that of cortex. However, this is very variable and, especially posteriorly, the high SI on T2W images may be quite marked when using FSE sequences.
The corpus callosum
The corpus callosum is thin, even in thickness (about 2mm) and fairly flat (Fig. 4.17a)5, 8, 49 . It is unmyelinated at term. The bulbous genu and splenium develop towards the end of the first year (Fig. 4.17b,c). It is of intermediate SI on T1W images but may be of low SI on T2W FSE images even though it is not myelinated (Fig. 4.3). The reason for this is unclear but it may be due to the compactness and uniformity of the fibers. However, the internal capsule, which is also made of densely packed fibers, does not have the same very low SI appearance on T2W FSE images.
THE BASAL GANGLIA AND THALAMI
The basal ganglia and thalami are relatively large in the term infant. The head of caudate nucleus, the lentiform nucleus (globus pallidus (GP) and putamen) and thalami are clearly demarcated by the internal capsule (Fig. 4.1e–h). The GP, VLNT and to a lesser extent the posterior putamen are prominent and of high SI on T1W images. The GP and the VLNT contain myelin from as early as 25 weeks 29, 32, 34, 36, 92 .
The GP on T1W images becomes less obvious in terms of SI from 38 to 42 weeks. The VLNT remains differentiated from the surrounding tissue for several months. The internal capsule, which is myelinated in its posterior third, lies between the putamen and the thalamus. Where the GP descends forwards to the upper mesencephalon it forms the anterior portion of a region in the diencephalon that has a very characteristic hair pin appearance (Fig. 4.1i). The central portion of the hair pin is of low SI on T1W images and consists of the unmyelinated tracts forming the internal capsule. The posterior portion is of high SI and is probably formed by the subthalamic nucleus.
THE HIPPOCAMPUS
The hippocampal region tends to be of high SI on T1W and low SI on T2W images but has a mixture of SIs with complicated cortical folding (Fig. 4.1 and Fig. 4.2). If the SI is uniform, especially high SI on T1W images, we consider that abnormal. It abuts on the anterior portion of the temporal horn of the lateral ventricle. A small area of myelination may be seen in this region at term on T1W images though the signal is difficult to differentiate from folded cortex.
THE BRAIN STEM
On T1W images the crura containing the pyramidal tracts begin to myelinate in their most lateral portions at term. The red nuclei and substantia nigra are of intermediate to low SI and often cannot be distinguished. The inferior and superior colliculi (the inferior colliculus is more prominent than the superior, (Fig. 4.4b,c) and the quadrigeminal plate, medial lemnisci and medial longitudinal fasciculi, and the decussation of the superior cerebellar peduncles are seen. All tend to be of high SI on T1 and low SI on T2W images. The optic chiasm is seen anteriorly (Fig. 4.1 and Fig. 4.2)8, 32, 59 79 .
The dorsal pons contains the myelinated medial and lateral lemnisci, the medial longitudinal fasciculi, and the middle cerebellar peduncles. All these structures are of high SI on T1W images and of low SI on T2W images. The ventral portion is generally of intermediate SI though there is some variation in SI in the pons indicating the beginning of MR differentiation of the tracts. Absence of this variation in SI is seen after severe perinatal asphyxia. The pons is prominent and measures about 11mm in antero-posterior diameter and 14.5mm in height53. The trigeminal nerve may be seen.
CEREBELLUM
The cerebellar hemispheres are of intermediate SI on T1W and T2W images (Figs 4.1–4.3) except for the dentate nuclei, the parasagittal portion of the vermis and the inferior and superior cerebellar peduncles which are of high SI on T1W and low SI on T2W images. In the more mature term infant the T1 and T2 begin to shorten. The middle cerebellar peduncles are not clearly demarcated from the body of the cerebellum79, 80. The vermis is about 20mm in antero-posterior diameter and 22.5mm in height53.
< prev | top | contents | next >
Contrast enhancement
Contrast enhancement is normally not seen or is only just apparent in the brain parenchyma, that is the cortex, white matter, basal ganglia and brain stem. However, obvious enhancement is seen in the pineal gland, the pituitary stalk, the dura, the large veins and sinuses and the choroid plexus in the lateral, third and fourth ventricles (Fig. 4.6). Barkovich et al7. note that enhancement is also seen in the metopic, coronal and lambdoid sutures, the diplo‘ of the basal skull bones and the iris, regardless of gestational age. Though studies on normal preterm infants are not reported, our own experience with a few infants is that the findings are similar to those reported for the term infant. When giving contrast we use dimeglumine gadopentetate (Magnevist; Schering Ltd, Burgess Hill, East Sussex, UK) in a dose of 0.2–0.4ml/kg intravenously.


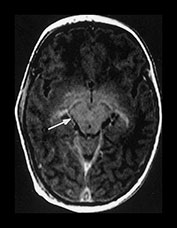


Fig. 4.6 Postcontrast images. Axial T1W (SE 860/30) contrast images at (a) high subcortical level showing enhancement in the falx (long arrow) and a cortical vein (short arrow); (b) basal ganglia level showing enhancement of the internal cerebral veins (long arrow) and the choroid plexus in the posterior horns of the lateral ventricles (short arrow); (c) the level of the mesencephalon showing enhancement in veins in the tentorial dura (white arrow); (d) the level of the fourth ventricle showing enhancement of the pituitary stalk (white arrow), the diplo‘ of the petrous temporal bone (short arrow) and the lambdoid suture (long arrow) as well as the transverse sinus; (e) the level of the medulla showing enhancement of the iris (short arrow) and choroid plexus in the foramina of Luschka (long arrow).
Diffusion weighted imaging of the neonatal brain
DIFFUSION WEIGHTED IMAGES
Diffusion weighted images (DWI) reflect the Brownian motion of water molecules within the brain. This diffusional motion is limited by tissue structure particularly in WM tracts where diffusion of water is freer along the length of axons than across them. Measurements of the diffusion of water therefore depend on the direction of the applied gradients in relation to the direction of the tract. The differences in diffusion in relation to axon (and tract) orientation can be seen on DWIs and is known as anisotropy. Normal WM shows anisotropy within myelinated and unmyelinated tracts. The tracts will appear of high SI if they are perpendicular to the direction of sensitization and of low SI if they are parallel to the direction of sensitization. The whole of the internal capsule shows anisotropy which is clearly seen, as does the external capsule, the corpus callosum, the optic radiation and other tracts within the cerebral hemispheres long before they are seen to be myelinated on T1 or T2W images. WM in the immediate periventricular region does not show the clear anisotropic features that are seen more centrally and more peripherally. Gray matter does not show anisotropy on visual inspection of images.
The clarity with which the tracts are seen is very dependent on the sequences used23, 66, 73, 82 , the number of data acquisitions and the immobility of the subject. The technique is very sensitive to motion artifact and the images are affected by the T2 weighting of the tissue. Details of the methodology we use are given in Table 4.1 and Refs 23 and 69 (Fig. 4.7). The problem of motion sensitivity, the variation in fiber direction and the proportion of fibers with long T2 components all affect the images and hence affect measurement of the apparent diffusion coefficient (ADC) of water in brain.
Abnormal tissue affected by an acute process often decreases its ADC and loses its anisotropic appearance so it becomes of uniform high SI irrespective of the direction of sensitization2, 23, 45, 47, 69, 72, 84 .




Fig. 4.7 Diffusion weighted images. DW axial images at the level of the basal ganglia sensitized in three directions: (a) anterior–posterior; (b) left–right; (c) head–foot (SE pulse interval/200ms, TR 1526–1572, b = 600s/mm2, four data acquisions); and (d) an IR image (3800/30/950) for comparison. Tracts at right angles to the direction of sensitization are of high SI and those parallel are of low SI. Note the clear change in SI in the internal capsule, corpus callosum, external capsule and tracts in the unmyelinated WM between (a), (b) and (c). No detail of the peripheral tracts is seen in (c) with the sensitization in the head–foot direction. The internal capsule is angulated at the genu and therefore the anterior and posterior portions (short and long arrows, c) are not of the same SI on any one image. The optic radiations seem to contain three tracts which are of high SI, low SI and high SI (short black arrows) on (a), all of high SI on (b), and of low SI and high SI on (c) and show very early myelination (arrow) on (d). The medial portion adjacent to the ventricle remains of high SI on all DW images. The anterior periventricular WM does not show clear anisotropic change (short arrow, b) and nor does the VLNT although it contains myelin and is of high SI in T1W images (d), of low SI on T2W images (not shown) and on all the DWIs (e.g. long arrow, b). This lack of anisotropy is probably due to mixed fiber orientation and short T2 effects.
APPARENT DIFFUSION COEFFICIENT MAPS
ADC maps of the normal neonatal brain show the corpus callosum, the optic radiation and the PLIC to be of low SI before they are seen to be myelinated on conventional imaging. Measurements of the ADC in both gray and white matter give higher values than those found in adults. Unlike the adult 70 ADCs are higher in WM than gray matter. There is also a strong correlation with gestational age with the ADCs of gray and hemispheric WM both decreasing and becoming closer to each other towards term44, 66, 68 . This is despite the fact that these fibers are not myelinated. The ADC in the PLIC also decreases with increasing gestational age 68. but according to Hüppi et al. the change is minimal though significant44.
The ADC in the PLIC is lower and nearer to that of the adult than the ADC of the ALIC reflecting the fact that myelination is more advanced in the PLIC. Similarly, at birth the ADC of occipital tracts is lower than the ADC of frontal tracts reflecting the caudo–cephalic progress of WM maturation. Measurements of the ADC within unmyelinated WM also show a rapid decrease over the first 6 post-term months65, 85. Values also decrease in frontal and occipital WM, the corpus callosum and the lentiform nucleus. The change in ADC is most marked when measured at 90 degrees to tracts. There is little change in ADCs with age when they are measured with gradients parallel to the tract66, 82.
Of note is the fact that the ADC in periventricular WM (but not the PLIC) seems to be higher in healthy preterm infants at term than in newborn full-term infants44. This suggests that periventricular WM is less mature in the preterm infant at term than the full-term infant and is in keeping with the observation that the T2 of this WM is long on conventional images42, 52, 57 . It is interesting that a difference is not seen in the PLIC which on conventional imaging is not delayed in its myelination in healthy premature infants, and indeed in our experience may be slightly advanced.
These observations show that the diffusion of water is less restricted in all tissues of the brain of the newborn than it is in the adult. It is well known that the fetal and infant brain contains more water than the brain of the older child and adult29 and that the water content reduces rapidly towards the end of gestation and in early infancy. The rapid changes in ADC during this time reflect this change in water content as well as localized restriction to the diffusional movement of water due to increasing numbers of oligo dendrocytes surrounding axons and later thickening of the myelin sheath and its reduced permeability to water. The changes during the time periods when measurements have been possible, that is the third trimester and in the early postnatal period, are greater frontally than occipitally and are concurrent with the caudo–cephalic progress of myelination. It appears that the WM in the hemispheres of preterm infants at term is less mature than it is in the full-term infants.
DIFFUSIONAL ANISOTROPY
If the entire explanation for anisotropy in WM was myelination of tracts, then one would expect to find that unmyelinated tracts did not show directional changes in ADC. This is not the case. In addition, measurements of quantitative diffusional anisotropy (DA) should inversely parallel the ADCs. Whilst the data from Hüppi et al.44 support this, the work from Neil et al.67 does not. Both groups have found that values of DA are greatest in the PLIC, lower in the ALIC (Neil et al.) and lower still in the centrum semiovale. Unlike Hüppi et al., Neil et al. found that measurements of DA in the PLIC, ALIC and corpus callosum did not change during the period from 30 to 40 weeks’ gestation. This is in contrast to the ADC values which gradually decreased with age in all tissues though Hüppi found the change in the PLIC to be small. Neil et al. found the DA to increase only within the unmyelinated WM of the centrum semiovale at the end of gestation. Vector maps of fiber orientation show orientation of unmyelinated fibers in the corpus callosum in the term infant. In the periventricular WM bundles of fibers are also seen but they are less tightly organised (Fig. 4.7). Again it is of note that the preterm infant at term has a lower DA in the hemispheric WM and the orientation of fibers seems less organized than in the full-term infant.
The cause of the anisotropy in the newborn brain is uncertain at present but it is becoming clearer. Anisotropic appearances are seen in histologically unmyelinated white matter90 and may be due to the premyelination state10 characterized by an increase in axon diameter and axonal membrane changes. This is accompanied by interactions between premyelinated axons and glial cells and by many other ultrastructural and functional changes.
The changes seen on DW images, in measurements of ADC and DA, and on vector maps in the third trimester and early postnatal period, particularly within periventricular WM, offer great potential for visualizing and understanding the processes of WM maturation. However, problems related to knowing the fiber orientation and proportion of long T2 components in the fibers affect all forms of DWI and make image interpretation difficult.
< prev | top | contents | next >
MR changes in the cerebrum, brain stem and cerebellum occurring after term:
conventional T1 and T2 weighted sequences
THE CORTEX
There is some increase in the depth and complexity of cortical folding just after term1, 12–14 and the Sylvian (lateral) fissure may close further, but there is little change that is obvious on conventional MR brain imaging. Of note, however, is the fact that the SI of the cortex which is high on T1W images and low on T2W images becomes of intermediate SI by 4–6 months. By this time the WM has myelinated and become of high SI on T1W and later low SI on T2W images. Therefore during the first few months after birth the images go through a phase of relative isointensity compared to earlier and later images. The exact timing of the changes in SI in gray matter and WM depends on the detail of the T1W and T2W sequences that are used. This reduction in SI in the cortex may be due to a reduction in cellularity and an increase in the number of synapses present at this time. It may therefore reflect increasing cortical complexity. Potentially, this could be measured from the changes in T1 and T2 of the tissue. Unfortunately, this is technically difficult as voxels containing only cortex have to be very small and placed in areas where the cortex is very folded to ensure exclusion of the adjacent WM.
As discussed above, the low SI on T2W images (particularly in the perirolandic region) persists for several months more than the high SI seen on T1W images.
WHITE MATTER
The changes in volume and myelination of the WM are dramatic16, 29, 34, 50, 92 . The WM provides the bulk of the brain volume and hence brain growth over the first 2 years. From 29–41 weeks’ gestation the rate of increase is 22ml/week43. This increase is accompanied by an increase in head circumference from 34–36cm at birth of about 0.5cm/week for the first 3 months and then at decreasing rates to 43–45cm at 1 year.
Volumetric growth of the brain can be measured from serially acquired images that are registered to each other and subtracted17, 76. In the adult this process results in an image of almost pure noise17, but in the infant because of growth and changes in the SI of tissue the subtraction image shows structure76. The increase in volume is seen as a rim of tissue around the circumference of the image and demonstrates that there is little change in the size of the basal ganglia and thalami. It also allows the process of myelination to be documented (e.g. along the corpus callosum) and can be used to demonstrate a decrease in T1 and T2 in WM prior to myelination.
Myelination occurs at different rates on T1W and T2W images. Before term myelin is best seen on FSE T2W images. After term for about 8–10 months myelin is better seen on T1W SE and more so on T1W IR images. After that time the major changes on T1W images have occurred and further maturation and detail is again better seen on T2W images.
As a general rule, myelination occurs in a caudal to cephalic direction and in the hemispheres is seen first parietally then occipital, frontally and temporally. The primary tracts myelinate before those in the association areas of the hemispheres. In the brain stem myelination occurs dorsally before ventrally, and in sensory before motor tracts.
Progress of myelination from term to 2 years
On T1W images the internal capsule is myelinated to the genu by 1 month (Fig. 4.8) and in its entirety by 3 months (Fig. 4.9). The splenium of the corpus callosum begins to myelinate at 3 months and the genu by 6 months (Fig. 4.17). The WM of the motor tracts are myelinated out to the subcortical regions by 3 months (Fig. 4.9) and to the primary visual regions of the occipital lobes by 6–7 months (Fig. 4.11). Myelin is seen in the frontal lobes by 5–6 months (Fig. 4.11) and in the temporal lobes by 7–8 months. Subcortical myelin appears in the frontal lobes at 8 months and temporally at 10 months (Fig. 4.13). Little change is seen on T1W images after this time though over the second year there is some extension of myelination in U fibers apparent in the subcortical WM (Fig. 4.15).

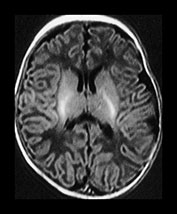

Fig. 4.8 Axial IR (IR 3800/30/950) images at 6 weeks. The PLIC is fully myelinated and the beginning of myelination are seen in the ALIC. Myelin is seen in the central motor tracts. The perirolandic cortex is of high SI.
On T2W images the changes are slower and more prolonged and hence subtle changes and delays in myelination in the second year are better demonstrated with T2W images. Myelination in the PLIC does not reach the genu before 4–6 months. It starts in the ALIC around 7 months and is not complete before 10 months depending on the particular T2W sequence used. Generally, it will appear sooner on FSE than on conventional SE images. The splenium of the corpus callosum becomes of low SI around 6 months and the genu by 8 months.
The WM of the motor tracts starts to become of low SI around 2 months and reaches the subcortical regions by 3–4 months. Low SI is seen in the optic radiations by 2 months and just extends to the calcarine fissure by 4 months. Other regions do not myelinate in terms of the T2 appearances before 9–12 months in the occipital lobes, 11–14 months frontally (Fig. 4.14) and 16–18 months in the temporal lobes. The low SI extends into all the subcortical regions by 2 years (Fig. 4.16).
Details of the timing of the appearance of myelin on T1W and T2W images are given in Table 4.4 and are well described by various authors 4, 8, 19, 28, 37–39, 58, 86–88 .
< prev | top | contents | next >
Table 4.4 Distribution of myelin seen on MR images from term to 2 years
| T1W images | T2W images |
|---|---|
| 1 month | |
| PLIC, optic radiation, central CS, | Posterior 1/3 PLIC, |
| optic nerve, cerebellar peduncles, | central CS, dorsal pons, |
| dorsal pons, vermis, | peridental WM, vermis |
| dentate nucleus | |
| 2 months | |
| Pre- and postcentral sulcus WM, | PLIC 1/3, CS, optic radiation, |
| ALIC, anterior pons, | middle cerebellar peduncles, |
| hippocampus | dentate nuclei |
| 3 months | |
| ALIC complete, | 1/2 PLIC |
| occipital WM at calcarine fissure, | |
| splenium of corpus callosum | |
| 4 months | |
| Beginning of frontal WM | Calcarine WM |
| 5 months | |
| Genu of corpus callosum | Anterior pons |
| 6 months | |
| Branching of frontal WM | PLIC complete, ALIC beginning, |
| splenium of corpus callosum | |
| 7 months | |
| Branching of occipital WM | Optic radiation complete |
| 8 months | |
| Peripheral frontal WM, | ALIC complete, |
| fine arborization of paracentral and | genu of corpus callosum |
| occipital WM | |
| 10 months | ALIC thick, |
| arborization of central motor tracts, | |
| branching of occipital tracts | |
| 11–12 months | |
| Temporal WM | Branching of posterior parietal tracts, |
| beginning of frontal tracts | |
| 15–18 months | Frontal arborization, |
| Subcortical WM, U fibers | beginning of temporal WM |
| 24 months | Temporal arborization, |
| subcortical WM, U fibers | |
The development of myelination described above is largely descriptive and allows for variations in images produced by different sequences at different field strengths. It is important to know both the general principles that govern the pattern and the rates of myelination of different regions of the brain. For detecting minor variations in myelination it is necessary to know the individual timing of changes in T1 and T2 in the sequences that are available on the particular system being used. We prefer to use FSE sequences up to term, IR sequences from term to 10 months and conventional T2W sequences to 2 years (Table 4.1). Gray and unmyelinated WM should be of almost equal SI at 4 months on T1W images. On T2W images the cortical and most of the central gray matter signal is isointense with myelinated WM around 1 year and the cortical signal becomes higher than myelinated WM between 18 months and 2 years. A very detailed description of the stages of myelination is given by van der Knaap and Valk88.
The terminal zones
Within myelinating WM there are regions of apparently unmyelinated WM. These are particularly obvious in the periventricular zones posterior and superior to the lateral ventricles (Fig. 4.16 and Fig. 4.18). They appear as patches of high SI within the low SI WM on T2W images and to a lesser extent regions of low SI within the high SI myelinated WM on T1W images. These regions are normal in infants in the second year and often persist for many years. They were described as ‘terminal zones’ by Yakovlev and Lecours92 because these regions do not stain for myelin for many years. They are felt to be normal but do occur in the region of WM most commonly affected in periventricular leukomalacia (PVL) which also produces areas of similar SI and needs to be distinguished from them. Their sites are very much those of the regions noted before term, and at term, within the WM that are of low SI on T1 and high SI on T2W images and correspond to cell-poor areas on histology32. These same regions commonly show patchy hyperintensity on T2W images in the elderly in whom on histology the myelin looks pale, there are dilated perivascular spaces, perivascular gliosis and ischemic change51.
In our experience these regions appear more obvious in very preterm infants without cerebral palsy who are examined at 2 years of age. They are also commonly seen in children imaged for global developmental delay for which no specific cause is found64. In these situations the ventricular margin is fairly smooth and of normal shape and there is a band of tissue of low SI on T2W images which separates the regions of long T2 from the ventricular margin.
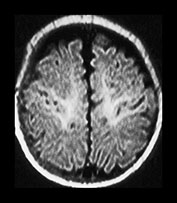


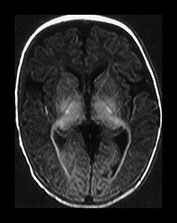
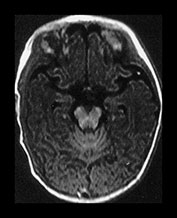
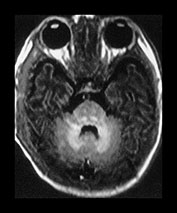
Fig. 4.9 Axial IR (IR 3400/30/800) images at 3 months. Myelination is seen in the subcortical motor tracts, the whole of the internal capsule, the splenium of the corpus callosum, the hippocampus, the optic radiations to the calcarine fissure, the cortico spinal tracts and deep cerebellar WM. The cortex around the central sulcus is no longer of high SI. The inferior portion of the GP and the VLNT are of relatively high SI.
< prev | top | contents | next >




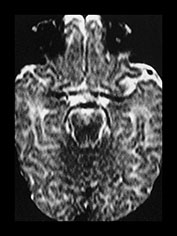

Fig. 4.10 Axial T2W (SE 2700/120) images at 3 months. Myelination is seen in the central periventricular WM, half of the PLIC and in the optic radiations. The cortex of the central sulcus is still of low signal intensity. The GP is of higher SI than the putamen and caudate nucleus.
< prev | top | contents | next >
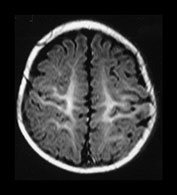








Fig. 4.11 Axial IR (IR 3400/30/800) images at 6 months. Myelination is almost mature in the central parietal lobes, it extends to the occipital subcortical WM, is present in the anterior and posterior portions of the parietal lobes and beginning to appear in the frontal lobes. The internal capsule has an irregular appearance due to fibers transversing it. The GP and VLNT are of higher SI than the rest of the basal ganglia and the posterior thalamus. The corticospinal tracts in the mesencephalon are well myelinated. The peri-aqueductal gray matter is now of low SI.
< prev | top | contents | next >

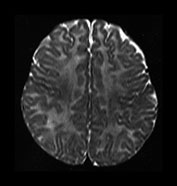
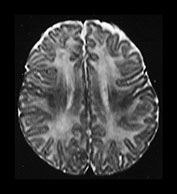
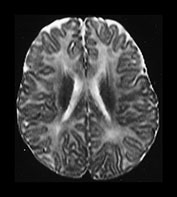




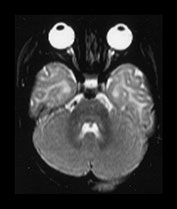
Fig. 4.12 Axial T2W (SE 2700/120) images at 6 months. Myelination is present in the central CS extending out to the subcortical WM of the central sulcus, the whole of the internal capsule, the splenium of the corpus callosum and the hippocampus. The GP and the posterior thalamus are of higher SI than the putamen and caudate nuclei and the VLNT remains of lower SI. The brain stem and cerebellar peduncles are also myelinated but no myelin is seen in the cerebellar folia.
< prev | top | contents | next >





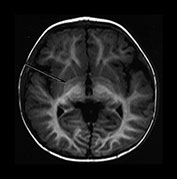


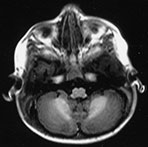
Fig. 4.13 Axial IR (IR 3400/30/800) images at 12 months. At this age myelination is apparent throughout the WM apart from the most peripheral arcuate fibers. In the basal ganglia the border of the GP is clearly demarcated and, as with the VLNT, the GP remains of higher SI than the adjacent tissue. The mamillary bodies are seen in the anterior thalami (d, short arrow). The anterior commissure is well seen (f, long arrow). Small cyst-like spaces are seen adjacent to the anterior horns of the lateral ventricles (d).
< prev | top | contents | next >



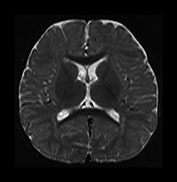
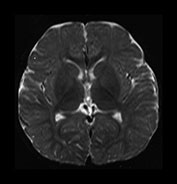

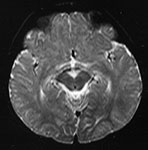


Fig. 4.14 Axial T2W (SE 2700/120) images at 12 months. There is more obvious low SI throughout the brain except for the infero-anterior portions of the temporal lobes. The cortex and myelinated WM are of similar SI at this age. The pons is clearly demarcated again with low SI in the ventral portion and intermediate signal dorsally.
< prev | top | contents | next >


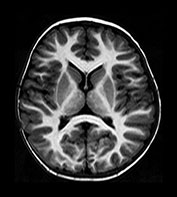
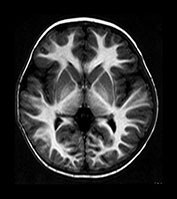

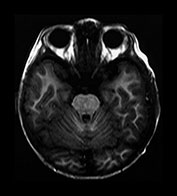
Fig. 4.15 Axial IR (IR 3100/30/700) images at 2 years. The brain has an adult appearance on these images with extensive subcortical myelination though some
U fibers are still to myelinate. In the posterior CS there are some patches of slightly lower SI within the myelinated WM.
< prev | top | contents | next >
In PVL associated with a motor deficit, the ventricles have an irregular margin and the high SI on the T2W images abuts on the ventricular margin, unlike the situation seen with normal terminal zones. Usually the high SI in PVL is more marked than is seen with normal terminal zones and with PVL there is usually an abnormal degree of thinning of at least the posterior portion of the body of the corpus callosum. This appearance can also be seen in a few children who had an acute encephalopathy at term with no evidence for preterm damage24.
Another differential diagnosis for regions of high SI and low SI on T2W and T1W images respectively are vascular spaces. These generally have a distinct linear appearance on the T1W images (see below, Fig. 4.22) and are best seen on the IR images. They can mimic abnormal long T2 in the WM if no T1 image is available.
The corpus callosum
The first postnatal changes that occur are a thickening of the genu and the splenium during the first 2–4 months with the genu being somewhat ahead of the splenium. However, myelination occurs first in the splenium around 4 months and in the genu around 6 months on T1W images. The body also thickens and takes on a more adult appearance around 9 months, often, though not always, with a region of focal thinning at the junction between the body and the splenium. The overall length of the corpus callosum seems to relate more to head size and shape than to the presence of disease5, 8, 49 .
White matter imaging with FLAIR sequences
Myelinated WM is well seen on fluid attenuated inversion recovery (FLAIR) images which have the advantage over T2W SE images of nulling the signal from CSF. We have not found FLAIR sequences to be of particular help in defining normal development in the young infant but in the second year of age interesting observations can be made. Characteristic regions of high SI are seen in the posterior portion of the PLIC, the optic radiations, particularly in their distal portions and adjacent to the frontal horns (Fig. 4.18). The PLIC and optic radiations are sites of early myelination and appear to retain long T2 components after myelination. The terminal zones, not surprisingly, are also seen to be of high SI but the cause for the high SI near the frontal horns is uncertain. It may have a common etiology with the cyst-like areas seen well on IR images in infants in the second year (Fig. 4.13) and the histological features noted amongst others by Dooling et al.30 and Leifer et al.55. Unmyelinated WM is also of high SI (Fig. 4.18a) with this sequence.
THE BASAL GANGLIA AND THALAMI
Between 37 and 42 weeks the high SI on T1W images in the GP and the posterior putamen diminishes considerably but the GP remains of higher SI (Figs 4.8, 4.9, 4.11, 4.13 and 4.15). It becomes clearly demarcated from the rest of the lentiform nucleus. The caudate nucleus remains of intermediate SI. On T2W images the region of the GP is of slightly increased SI throughout the first year. The posterior putamen loses the low SI seen in the neonatal period and becomes of intermediate SI like the caudate nucleus.
The high SI in the VLNT seen on T1W images in the neonatal period persists for longer than the marked high SI in the GP and remains to some extent through the second year (Fig. 4.15). On T2W images the VLNT is of low SI neonatally and remains so, though to a lesser extent through to the second year. The pulvinar of the thalamus is of lower SI on T1W and higher SI on T2W images than the more anterior thalamus.
The iron deposition which reduces the SI of the GP, substantia nigra and red nucleus (and later the dentate nucleus) on T2W images is not seen before 9 years of age.
THE HIPPOCAMPUS
Whilst there may be a very small area of myelinated tissue at term, myelination is always seen by 3 months (Fig. 4.9) and the region is well myelinated on T1W images by 12 months (Fig. 4.13). It is slow to myelinate fully on T2W images even by age 2 years (Fig. 4.16). The overall appearance of this region of the temporal lobe on axial views is of a thick band of folded tissue of mixed SIs lying medial to the temporal horns. The hippocampus is often better seen in the coronal rather than the axial or sagittal planes.

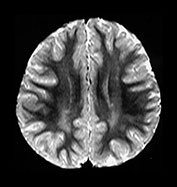
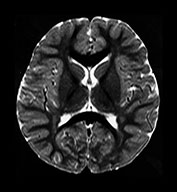

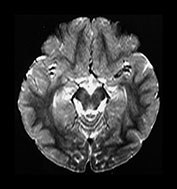

Fig. 4.16 Axial T2W (SE 2700/120) images at 2 years. Myelination has progressed throughout all lobes but has not extended as far in the frontal and temporal lobes as it has on the IR images. Within the myelinated WM are regions of long T2 mainly in the trigones and postero-lateral to the posterior horns of the ventricles. These regions are called terminal zones and may take some years to myelinate fully.



Fig. 4.17 Sagittal T1W (SE 860/20) images of the corpus callosum at: (a) term showing a complete, thin, even and unmyelinated corpus callosum; (b) 4 months showing thickening of the genu and the posterior body and splenium, but only the latter portions are beginning to myelinate; (c) at 10 months showing thickening throughout with some residual thinning of the body at the junction between the posterior portion and the splenium.



Fig. 4.18 Axial FLAIR (8142/150/2100) images through: (a) cortex and subcortical WM; (b) high-ventricular level; (c) the basal ganglia. Myelinated WM and CSF are of low SI. Unmyelinated WM is best seen in (a) and is of high SI (arrow). High SI is seen in (b) in the posterior periventricular WM of the terminal zones (arrow). Regions of high SI are typically seen on FLAIR images adjacent to the anterior horns (long arrow) and in the optic radiations (short arrow, c).
< prev | top | contents | next >
THE BRAIN STEM
From birth to 3 months tracts and nuclei in the brain stem become increasingly obvious particularly on T1W images. The main features of these areas are shown in Fig. 4.19 taken from a 2-month-old infant.
CEREBELLUM
The inferior and superior cerebellar peduncles are already myelinated by term and the middle cerebellar peduncles myelinate in the first 2–3 months. By 3 months high SI is seen in the dentate nucleus, peridental WM and the corpus medullare and the appearances in these regions are similar to those seen in the adult. Myelination on T2W images takes a little longer. More peripheral myelination in the cerebellum extending to the folia begins to appear after 4 months on T1W images and after 8 months on T2W images and the cerebellum has an adult appearance by 18 months (Figs 4.9–4.19)8 39, 80, 86, 88 .
< prev | top | contents | next >
Observations of other areas of the brain seen on MR brain scans from term to 2 years
THE PITUITARY GLAND
The pituitary gland is well seen on T1W mid-line sagittal 4mm thick slice images8, 25, 91 . The anterior portion is of high SI up to 2 months of age. The posterior portion is also of high SI but may not always be seen separate from the anterior portion. Thereafter, the anterior portion is of lower SI than the posterior portion and is easier to differentiate. The gland is described25 as being upwardly convex for this period. It gradually becomes flatter in most, but not all, infants though in our experience this is not always the case. It also increases in length slightly from 6 to 7 mm over the first year but the height remains the same at about 3.5–4mm. It does not grow as fast as the rest of the brain over the first year. The size of the stalk does not change and it should never be as large as the basilar artery. The pituitary infundibulum enhances with contrast.
THE PINEAL GLAND
The pineal gland develops from the caudal portion of the roof of the third ventricle. At birth it has a volume of about 15–40 mm3 and increases to about twice that size by 2 years. It can be seen on very thin 1mm T1W sagittal or coronal images lying just above the quadrigeminal plate between the thalami and beneath the junction of the posterior body and the splenium of the corpus callosum (Fig. 4.21)81. It does not alter in size after the first year78.
THE VENTRICULAR SYSTEM
The lateral ventricles are small and rounded anteriorly and posteriorly (Figs 4.1–4.3) and are clearly seen extending into the temporal lobes. McArdle et al.62 studied the size of the ventricles in the axial plane in relation to brain diameter. They suggested that the normal ratio of lateral ventricle/ brain lies between 0.29 and 0.32 at term at the level of the anterior horns and at mid-ventricular level. The third ventricle is just seen posteriorly but the anterior portion may be more obvious and dips antero-inferiorly towards the pituitary fossa. The aqueduct is narrow but visible (Figs 4.1j and 4.2d) The fourth ventricle is slightly wider than it is deep, and about half the diameter of the pons (Fig. 4.1 and Fig. 4.19).


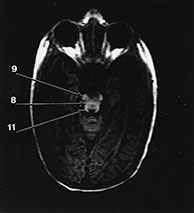




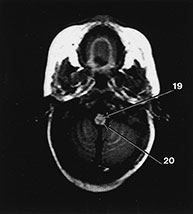
Fig. 4.19 Brain stem. Axial IR (IR 3800/30/950) images through the brain stem, occipital lobes and cerebellum of a 2-month-old infant. 1, optic tract; 2, optic radiation; 3, hippocampus; 4, aqueduct; 5, lateral geniculate body; 6, medial longitudinal fasciculus; 7, medial lemniscus; 8, decussation of the superior cerebellar peduncles; 9, myelinated corticospinal tracts; 10, inferior colliculus; 11, lateral lemniscus; 12, pyramidal tracts; 13, superior cerebellar peduncle; 14, 4th ventricle; 15, trigeminal nerve; 16, middle cerebellar peduncle; 17, inferior cerebellar peduncle; 18, olive; 19, decussation of the pyramidal tracts; 20, gracile and cuneate nuclei. (a)–(c) mesencephalon; (d) –(e) pons; (f)–(g) medulla; (h) brain stem.


Fig. 4.20 Pituitary gland. Sagittal TIW images (860/20) showing the pituitary gland at (a) term and (b) 2 months post-term. The gland is initially of uniform high SI usually with an upward convexity. By 2 months the anterior pituitary is of intermediate SI.


Fig. 4.21 Pineal gland. Sagittal T1W (SE 860/20) images showing the pineal gland marked by an arrow (a) at term and
(b) at 2 years.
THE EXTRACEREBRAL SPACES
These spaces are best assessed on T2W images where the outer cortical margin and the inner table of the skull can be clearly differentiated from CSF62.
The interhemispheric fissure is narrow and regular (Fig. 4.1 and Fig. 4.3) but the Sylvian fissures may be prominent. The depth of the insular cortex is 6–10mm in 35–42 weeks’ gestation infants. The width of the subarachnoid space ranges from 0 to 3mm, being generally 0–3mm over the convexities and in the interhemispheric fissure and 2–3mm in the ambient cisterns. The space around the parietal and temporal lobes and the basilar cisterns may appear quite large. It is generally 2–4mm in the cerebello-pontine angle cistern but the space anterior to the temporal lobe in the middle fossa and anterior to the frontal lobes may be up to 5–6mm. Some apparently normal term infants may have very wide subarachnoid spaces as large as 12–13mm posterior to the parietal lobes. In our experience this is an infrequent observation at term but may be a normal finding up to about 36–37 weeks. We see it in almost all ex-premature infants at this age. Enlarged frontal spaces are not uncommon and may be associated with normal development in children scanned because of macrocephaly26. A large cisterna magna may be a normal variant but care must be taken to ensure that the cerebellum, in particular the vermis, are of normal size.
VASCULAR SPACES
Vascular spaces are common in the elderly, but they can be seen in the newborn within the tissue of the inferior basal ganglia near the lateral portions of the anterior commissure (Fig. 4.1i and Fig. 4.22c) They usually have a linear shape, and are angled anteriorly and towards the mid-line. There may be several. Care needs to be taken to recognize abnormal vascular spaces, for example in Leigh’s disease where the cysts are usually larger and more superior in their distribution.
In the older child regions of long T1 and T2 are seen within the myelinated white matter usually in a periventricular distribution (Fig. 4.22a–d) These have a linear appearance and are also thought to be vascular spaces. They are generally not seen before the WM in the hemispheres is well myelinated. Similar areas, usually smaller and more rounded, can be seen in the basal ganglia (Fig. 4.22b).
Small spaces of cyst-like appearance are common adjacent and antero-lateral to the anterior horns (Fig. 4.22b,d). These have not been described in the literature and their etiology is not clear. They may be remnants of subependymal cysts that are not seen until the frontal periventricular WM is well myelinated.
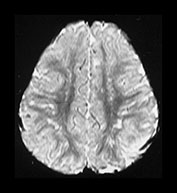
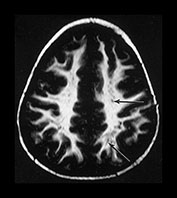
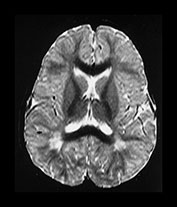

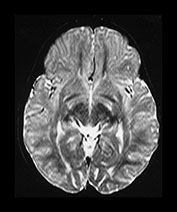

Fig. 4.22 Vascular spaces. Axial IR images T2W (SE 3000/120) (i) and (IR 3800/30/950) (ii) at the level of (a) the centrum semiovale; (b) the basal ganglia; and (c) the upper mesencephalon. Characteristic small elongated regions of low SI on the IR and high SI on the T2W images are seen (arrows) within myelinated WM (a,b), within the lentiform nucleus (b) and adjacent to the lateral portions of the anterior commissure (c). In addition there are similar regions adjacent to the anterior horns which are of uncertain etiology.
DEVELOPMENTAL SPACES
The cavum septum pellucidum and cavum vergum are still patent in at least 75% of term infants but close rapidly. The cavum septum pellucidum is patent in 6% of children and 1% of adults11. Persistance of a large cavum septum pellucidum may be seen as part of the spectrum of mid line developmental anomalies.
< prev | top | contents | next >
Functional MR imaging
The subject is covered in more detail in Chapter 16. Studies using functional MR imaging (fMRI) techniques in normal infants are scarce. Most adult studies in this field require the active co-operation of the subject combined with the ability to lie very still, which is clearly not possible in this young age group. Approaches have been limited to studying sensory systems where the stimulus will not disturb the infant. Few entirely normal infants have been studied and none in the awake state.
fMRI attempts to map the distribution of neuronal activity that is involved in a particular active or passive process. It is not a direct measure of neuronal activity but measures changes to local cerebral blood volume, flow or blood oxygenation associated with neuronal activity. The most common imaging method used has the acronym BOLD (blood oxygenation level dependent). Usually spin echo echo-planar sequences with a long TR and long TE or gradient echo sequences are used. The former are less sensitive to motion artifact but problems with motion artifact remain an issue with fMRI35.
The technique depends largely on detecting changes in local levels of deoxygenated hemoglobin. Cortical venous and capillary blood has a relatively high concentration of deoxygenated hemoglobin. In response to neuronal activation there is a rapid increase in local blood flow which is in excess of that needed to supply oxygen for the increased metabolic demand. This results in a decrease in deoxygenated hemoglobin in the local capillary and venous blood. As deoxyhemoglobin is paramagnetic there is a change in local SI which can be detected by subtraction of pre-stimulus from post-stimulus images. In general, in adult studies, the excess of oxyhemoglobin and decrease in deoxyhemoglobin in the post-stimulus phase results in an increase in SI and therefore a positive BOLD effect.
The studies in newborns and young infants have concentrated on the visual system which can be stimulated in the sleeping and sedated child without disturbance. The visual system is complicated to study. It is thought that the young infant does not use the primary occipital cortex for vision but a subcortical system involving the pulvinar and the superior colliculi3, 31 48 . The infant may only have obligate use of the occipital cortex for vision after 2–4 months.
The results of infant studies have been difficult to interpret as positive, negative and absent responses have been found in the primary visual area in infants with apparently normal visual systems12–14 60 93 . When the BOLD contrast signal was negative it was often not in the primary visual area but anterior and lateral to it, though in the first month after term a positive BOLD signal was sometimes seen in there. There are probably many explanations for the variation in findings which may partly reflect the processing changes in the visual system in the first months after birth or possibly a relative excess of synapses at this age93 as compared with later in the first year. Other factors are likely to be non-optimal stimulus paradigms, smaller changes in local blood flow, volume or oxygenation in response to a stimulus than are found in the older child and adult, stimulation of the visual system through closed eyelids, and the effects of sleep or of sedation46 or motion artifact.
fMRI is an exciting aspect of brain imaging that is only beginning to be applied to the pediatric population. fMRI has shown sites of activity in response to visual stimulation in brains affected by congenital and acquired abnormality of the occipital lobes14, 56. It is hard to see at present how studies of the normal child in the awake state can be achieved until they are old enough to co-operate. However, the studies of the visual system are encouraging and it should be possible to perform similar work with the auditory and cutaneous systems.
< prev | top | contents | next >
Summary
- MRI gives superb detail of the developing brain. It is essential to have a thorough knowledge of the normal appearances at different ages in order to define the abnormal.
- Structures may look very different depending on the strength of field, the sequence, the plane, and the angle of acquistion. Normal appearances have to be established with the advent of any new MR technique.
- Myelinated structures have a short T1 and short T2. In our experience, prior to term these are best seen using a T2 weighted fast spin echo sequence between term and 1year using an inversion recovery sequence and after 1year a T2 weighted spin echo sequence.
- A T1 weighted or inversion recovery sequence in the transverse plane is invaluable in the assessment of the neurologically abnormal neonate at term.
- Diffusion weighted imaging allows definition of white matter tracts prior to MR visible myelination and even prior to histological myelination.
- Volumetric growth of the brain and its intrinsic structures can be objectively quantified using serially acquired images.
- Whilst the normal appearances and sizes of some structures are known, the clinical significance of mild deviations from the normal are seldom well understood.
< prev | top | contents | next >
Abbreviations
| ADC | apparent diffusion coefficient |
| ALIC | anterior limb of the internal capsule |
| b | diffusion sensitivity parameter |
| b/w | black and white |
| BOLD | blood oxygen level dependent |
| CS | centrum semiovale |
| CT | computed tomography |
| DA | diffusional anisotropy |
| DWI | diffusion weighted imaging |
| FLAIR | fluid attenuated inversion recovery |
| fMRI | functional magnetic resonance imaging |
| FOV | field of view |
| FSE | fast spin echo |
| GP | globus pallidus |
| IR | inversion recovery |
| MR | magnetic resonance |
| PLIC | posterior limb of the internal capsule |
| PVL | periventricular leukomalacia |
| SE | spin echo |
| SI | signal intensity |
| T | Tesla |
| TE | echo time |
| TI | inversion time |
| TR | repetition time |
| T1W | T1 weighted |
| T2W | T2 weighted |
| VLNT | ventro-lateral nucleus of the thalamus |
| WM | white matter |
< prev | top | contents | next >
References
- Ajayi-Obe M, Saeed N, Cowan FM et al. (2000) Reduced development of cerebral cortex in extremely preterm infants. Lancet 356, 1162–1163.
- Arbelaez A, Castillo M and Mukherji SK (1999) Diffusion-weighted MR imaging of global cerebral anoxia. Am J Neuroradiol 20, 999–1007.
- Atkinson J (1992) Early visual development: differential functioning of parvocellular and magnocellular pathways. Eye 6, 129–135.
- Baier P, Forster Ch, Fendel H et al. (1998) Magnetic resonance imaging of normal and pathological white matter maturation. Pediatr Radiol 18, 183–189.
- Barkovich AJ and Kjos BO (1998) Normal postnatal development of the corpus callosum as demonstrated by MR imaging. Am J Neuroradiol 9, 487–491.
- Barkovich AJ, Kjos BO, Jackson DE et al. (1998) Normal maturation of the neonatal and infant brain: MR imaging at 1.5T. Radiology 166, 173–180.
- Barkovich AJ, Latal-Hajnal B, Partridge JC et al. (1997) MR contrast enhancement of the normal neonatal brain. Am J Neuroradiol 13, 1713–1717.
- Barkovich AJ (2000) Normal development of the neonatal or infant brain, skull and spine. In: Pediatric Neuro-imaging, 3rd edn. Philadelphia: Lippincott Williams & Wilkins, pp. 28–45.
- Becker L, Armstrong DL and Wood CF (1984) Dendritic development in human occipital cortical neurons. Dev Brain Res 13, 117–124.
- Black JA, Waxman SG, Ransom BR et al. (1986) A quantitative study of developing axons and glia following altered gliogenesis in rat optic nerve. Brain Res 380, 122–135.
- Bodensteiner JB, Schaefer GB and Craft JM (1998) Cavum septi pellucidi and cavum vergae in normal and developmentally delayed populations. J Child Neurol 13, 120–121.
- Born P, Rostrup E, Leth H et al. (1996) Change of visually induced cortical activation pattern during development. Lancet 347, 543.
- Born P, Leth H, Miranda M et al. (1998) Visual activation in infants and young children studied by functional magnetic resonance imaging. Pediatr Res 44, 578–583.
- Born AP, Miranda MJ, Rostrup E et al. (2000) Functional magnetic resonance imaging of the normal and abnormal visual system in early life. Neuropediatrics 31, 24–32.
- Boyko OB, Burger PC, Shelburne JD et al. (1992) Non-heme mechanisms for T1 shortening: pathologic, CT, and MR elucidation. Am J Neuroradiol 13, 143–145.
- Brody BA, Kinney HC, Kloman AS et al. (1987) Sequence of central nervous system myelination in human infancy. I. An autopsy study of myelination. J Neuropathol Exp Neurol 46, 283–301.
- Bydder GM (1995) Detection of small changes to the brainwith serial magnetic resonance imaging. Br J Radiology 68, 1271–1295.
- Childs A-M, Ramenghi LA, Evans DJ et al. (1998) MR featuresof developmental periventricular white matter in preterminfants: evidence of glial cell migration. Am J Neuroradiol 19, 971–976.
- Christophe C, Muller MF, Baleriaux D et al. (1990) Mapping of normal brain maturation in infants on phase-sensitive inversion-recovery MR images. Neuroradiology 32, 173–178.
- Conel JL (1941) The Postnatal Development of the Human Cerebral Cortex, Vol. II. The Cortex of the One-Month Infant. Cambridge, MA, Harvard University Press.
- Conel JL (1947) The Postnatal Development of the Human Cerebral Cortex, Vol. III. The Cortex of the Three-Month Infant. Cambridge, MA, Harvard University Press.
- Conel JL (1951) The Postnatal Development of the Human Cerebral Cortex, Vol. IV. The Cortex of the Six-Month Infant. Cambridge, MA, Harvard University Press.
- Cowan FM, Pennock J, Hanrahan JD et al. (1994) Early detection of cerebral infarction and hypoxic ischemic encephalopathy in neonates using diffusion-weighted magnetic resonance imaging. Neuropediatrics 25, 172–175.
- Cowan FM (2000) Outcome after perinatal asphyxia in the term infants. Johnson A (Ed.) Semin Neonatol 5, 1–14.
- Cox TD and Elster AD (1991) Normal pituitary gland: changes in shape, size and signal intensity during the 1st year of life at MR imaging. Radiology 179, 721–724.
- de Vries LS, Smet M, Ceulemans B et al. (1990) The role of high resolution ultrasound and MRI in the investigation of infants with macrocephaly. Neuropediatrics 21, 72–75.
- de Vries LS, Groenendaal F, van Haastert IC et al. (1999) Asymmetrical myelination of the posterior limb of the internal capsule in infants with periventricular haemorrhagic infarction; an early predictor of hemiplegia. Neuropediatrics 30, 314–319.
- Dietrich RB, Bradley WG, Zaragoza IV EJ et al. (1988) MR evaluation of early myelination pattern in normal and developmentally delayed infants. Am J Neuroradiol 9, 69–76.
- Dobbing J and Sands J (1973) Quantitative growth and development of human brain. Arch Dis Child 48, 757–767.
- Dooling EC, Chi JG and Gilles FH (1983) Developmental changes in ventricular epithelia. In: Gilles FH, Leviton A and Dooling EC (Eds) The Developing Human Brain: Growth and Epidemiologic Neuropathology. Boston, John Wright, pp. 113–116.
- Dubowitz LM, Mushin J, de Vries LS et al. (1986) Visual function in the newborn infant: is it cortically mediated? Lancet 1, 1139–1141.
- Feess-Higgins A and Larroche J-C (1987) Development of the human foetal brain – an anatomical atlas. Inserm CNRS.
- Felderhof-Meuser U, Maalouf E, Squier MV et al. (1999) Magnetic resonance imaging of the brain in very preterm infants: correlation with histopathological findings. Am J Neuroradiol 20, 1349–1357.
- Gilles FH, Shankle W and Dooling EC (1983) Myelinatedtracts: growth patterns. In: Gilles FH, Leviton A and Dooling EC (Eds) The Developing Human Brain. Boston, John Wright, pp. 117–192.
- Hajnal JV, Myers R, Oatridge A et al. (1994) Artefacts due to stimulus correlated motion in functional imaging of the brain. Magn Reson Med 31, 283–291.
- Hasegawa M, Houdou S, Mito T et al. (1992) Development of myelination in the human fetal and infant cerebrum: a myelin basic protein immunohistochemical study. Brain Dev 14, 1–6.
- Hayakawa K, Konishi Y, Kuriyama M et al. (1990) Normal brain maturation on MRI. Eur J Radiol 12, 208–215.
- Hittmair K, Wimberger D, Rand T et al. (1994) MR assessment of brain maturation: comparison of sequences. Am J Neuroradiol 151, 425–433.
- Hittmair K, Kraker J, Rand T et al. (1996) Infratentorial brain maturation: a comparison of MRI at 0.5 and 1.5 T. Neuroradiology 38, 360–366.
- Hock A, Demmel H, Schicha K et al. (1975) Trace element concentration in human brain. Brain 98, 49–64.
- Holland BA, Haas DK, Norman D et al. (1986) MRI of normal brain maturation. Am J Neuroradiol 71, 201–208.
- Hüppi PS, Schuknecht B, Boesch C et al. (1996) Structural and neurobehavioral delay in postnatal brain development of preterm infants. Pediatr Res 39, 895–901.
- Hüppi PS, Warfield S, Kikinis R et al. (1998) Quantitative magnetic resonance imaging of brain development in premature and mature newborns. Ann Neurol 43, 224–235.
- Hüppi PS, Maier SE, Peded S et al. (1998) Microstructural development of human newborn cerebral white matter assessed in vivo by diffusion tensor magnetic resonance imaging. Pediatr Res 44, 584–590.
- Inder T, Hüppi PS, Zientara GP et al. (1999) Early detection of periventricular leukomalacia by diffusion-weighted magnetic resonance imaging techniques. J Pediatr 134, 631–634.
- Joeri P, Huisman T, Loenneker Th et al. (1996) Reproducibility of fMRI and effects of pentobarbital sedation on cortical activation during visual stimulation. Neuroimage Suppl 3, S280.
- Johnson AJ, Lee BCP and Lin V (1999) Echoplanar diffusion-weighted imaging in neonates and infants with suspected hypoxic-ischaemic injury. Am J Roentgenol 172, 1721–1726.
- Johnson MH (1990) Cortical maturation and the development of visual attention in early infancy. J Cogn Neurosci 2, 81–95.
- Kier EL and Truwit CL (1996) The normal and abnormal genu of the corpus callosum: an evolutionary, embryologic, anatomic and MR analysis. Am J Neuroradiol 17, 1631–1641.
- Kinney HC, Brody BA, Kloman AS et al. (1987) Sequence of central nervous system myelination in human infancy. II. Patterns of myelination in autopsied infants. J Neuropathol Exp Neurol 47, 217–234.
- Kitajima M, Korogi Y, Okuda T et al. (1999) Hyperintensities of the optic radiation on T2-weighted MR images of elderly subjects. Am J Neuroradiol 20, 1009–1014.
- Koenig SH, Brown RD III, Spiller M et al. (1990) Relaxometry of the brain: why white matter appears bright on MR. Magn Reson Med 14, 482–495.
- Konishi Y, Hayakawa K, Kuriyama M et al. (1993) Developmental features of the brain in preterm and fullterm infants on MR imaging. Early Hum Dev 34, 155–162.
- Korogi Y, Takahashi M, Sumi M et al. (1996) MR signal intensity of the perirolandic cortex in the neonate and infant. Neuroradiology 38, 578–584.
- Leifer D, Buonanno FS and Richardson Jr EP (1990) Clinicopathological correlations of cranial magnetic resonance imaging of periventricular white matter. Neurology 40, 911–918.
- Lui WL, Biervert K, Hallmann A et al. (2000) Functional MRI in children with congenital structural abnormalities of the occipital cortex. Neuropediatrics 31, 13–15.
- Maalouf E, Duggan P, Rutherford MA et al. (1999) Magnetic resonance imaging of the brain in extremely preterm infants: normal and abnormal findings from birth to term. J Pediatrics 135, 351–357.
- Martin E, Kikinis R, Zuerrer M et al. (1988) Developmental stages of human brain: an MR study. J Comput Assist Tomogr 12, 917–922.
- Martin E, Krassnitzer S, Kaelin P et al. (1991) MR imaging of the brainstem: normal postnatal development. Neuroradiology 33, 391–395.
- Martin E, Joeri P, Leonneker T et al. (1999) Visual processing in infants and children studied using functional MRI. Pediatric Res 46, 135–140.
- McArdle CB, Richardson CJ, Nicholas DA et al. (1987) Developmental features of the neonatal brain: MR imaging. Part 1. Gray–white matter differentiation and myelination. Radiology 162, 223–229.
- McArdle CB, Richardson CJ, Nicholas DA et al. (1987) Developmental features of the neonatal brain: MR imaging. Part 2 Ventricles and extracerebral spaces. Radiology 162, 230–234.
- Mercuri E, Rutherford M, Cowan F et al. (1999) Early prognostic indicators in infants with neonatal cerebral infarction: a clinical, EEG and MRI study. Pediatrics 103, 39–46.
- Miyazaki M, Hashimoto T, Tayama M et al. (1992) Occipital deep white matter hyperintensity as seen by MRI: 1. Clinical significance. Brain Dev 14, 150–155.
- Morriss MC, Zimmerman RA, Bilaniuk LT et al. (1999) Changes in brain water diffusion during childhood. Neuroradiology 41, 929–934.
- Nomura Y, Sakuma H, Takeda K et al. (1994) Diffusional anisotropy of the human brain assessed with diffusion-weighted MR: relation with normal brain development and aging.Am J Neuroradiol 15, 231–238.
- Neil JJ, Shiran SI, McKinstry RC et al. (1998) Normal brain in human newborns: apparent diffusion coefficient and diffusion anisotropy measured by using diffusion tensor MR imaging. Radiology 209, 57–65.
- Oatridge A, Cowan F, Schwieso J et al. (1995) Age related changes in the apparent diffusion coefficient of white and grey matter in the normal infant brain. SMRM Abst., p. 1282.
- Pennock JM, Cowan FM, Schweiso JE et al. (1994) Clinical role of diffusion weighted imaging: neonatal studies. MAGMA 2, 273–278.
- Pierpaoli C, Jezzard P, Basser PJ et al. Diffusion tensor imaging of the human brain. Radiology 201, 637–648.
- Prenger ER, Beckett WW, Kollias SS et al. (1994) Comparison ofT2-weighted spin-echo and fast spin-echo techniques in the evaluation of myelination. J Magn Res Imaging 4, 179–184.
- Robertson RL, Ben-Sira L, Barnes PD et al. (1999) MR line-scan diffusion-weighted imaging of term neonates with perinatal brain ischemia. Am J Neuroradiol 20, 1658–1670.
- Rutherford M, Cowan F, Manzur AY et al. (1991) MR imaging of anisotropically restricted diffusion in the brain of neonates and infants. J Comput Assist Tomogr 15, 188–198.
- Rutherford MA, Pennock JM, Schwieso JE et al. (1995) Hypoxic ischaemic encephalopathy: early magnetic resonance imaging findings and their evolution. Neuropediatrics 26, 183–191.
- Rutherford MA, Pennock J, Schweizo JE et al. (1996) Hypoxic–ischaemic encephalopathy: early and late magnetic resonance imaging findings in relation to outcome. Arch Dis Child 75, F145–151.
- Rutherford MA, Pennock J, Cowan FM et al. (1997) Detection of subtle changes in the brains of infants and children via subvoxel registration and subtraction of serial MR images. Am J Neuroradiol 18, 829–835.
- Rutherford MA, Pennock J, Counsell S et al. (1998) Abnormal magnetic resonance signal in the internal capsule predicts poor neurodevelopmental outcome in infants with hypoxic–ischaemic encephalopathy. Pediatrics 202, 323–328.
- Schmidt F, Penka B, Trauner M et al. (1995) Lack of pineal growth during childhood. J Clin Endocrinol Metab 80, 1221–1225.
- Sie LT, van der Knaap MS, van Wezel-Meijler G et al. (1997) MRI assessment of myelination of motor and sensory pathways in the brain of preterm and term-born infants. Neuropediatrics 28, 97–105.
- Stricker T, Martin E and Boesch C (1990) Development of the human cerebellum observed with high-field-strength MR imaging. Radiology 177, 431–435.
- Sumida M, Barkovich AJ and Newton TH (1996) Development of the pineal gland: measurement with MR. Am J Neuroradiol 17, 233–236.
- Takeda K, Nomura Y, Sakuma H et al. (1997) MR assessment of normal brain development in neonates and infants: comparative study of T1- and diffusion-weighted images. J Comput Assist Tomogr 21, 1–7.
- Tanaka S, Mito T and Takashima S (1995) Progress of myelination in the human fetal spinal nerve roots, spinal cord and brainstem with myelin basic protein immunohistochemistry. Early Hum Dev 41, 49–59.
- Thornton JS, Ordidge RJ, Penrice J et al. (1998) Temporal and anatomical variations of brain water apparent diffusion coefficient in perinatal cerebral hypoxic–ischemic injury: relationships to cerebral energy metabolism. Magn Reson Med 39, 920–927.
- Tofts PB, Leth H, Peitersen B et al. (1996) The apparent diffusion coefficient of water in gray and white matter of the infant brain.J Comput Assist Tomogr 20, 1006–1011.
- van der Knaap MS and Valk J (1990) MR imaging of the various stages of normal myelination during the first year of life. Neuroradiology 31, 459–470.
- van der Knaap MS and Valk J (1991) Myelination as an expression of the functional maturity of the brain. Dev Med Child Neurol 33, 849–857.
- van der Knaap MS and Valk J (1995) Magnetic Resonance of Myelin, Myelination and Myelin Disorders, 2nd edn. Berlin, Springer, pp. 1–17 and 31–52.
- van Wezel-Meijler G, van der Knaap MS, Sie LTL et al. (1998) Magnetic resonance imaging of the brain in premature infants during the neonatal period. Neuropediatrics 29, 89–96.
- Wimberger DM, Roberts TP, Barkovich AJ et al. (1995) Identification of ‘premyelination’ by diffusion-weighted MRI. J Comput Assist Tomogr 19, 28–33.
- Wolpert XM, Osborne M, Anderson M et al. (1988) Bright pituitary gland: a normal MR appearance in infancy. Am J Neuroradiol 9, 1–3.
- Yakovlev PI and Lecours AR (1967) The myelogenetic cycles of regional maturation of the brain. In: Minkowski A (Ed.) Regional Development of the Brain in Early Life. Oxford, Blackwell, pp. 3–70.
- Yamada H, Sadato N, Konoshi Y et al. (1997) A rapid brain metabolic change in infants detected by fMRI. Neuroreport 8, 3775–3778.